TEG (THOMROBOELASTOGRAPH) PANEL
|
Geisinger Epic Procedure Code: LAB3774
Geisinger Epic ID: 140937
|
SPECIMEN COLLECTION |
Specimen type: |
Whole Blood |
Preferred collection container: |
|
Alternate Collection Container: |
Other size blue-top (3.2% sodium citrate) tubes (e.g., 1.8 mL, 4.5 mL)
|
Specimen required: |
One (1) full 2.7 mL light blue top (3.2% sodium citrate) tube of whole blood. |
Special notes: |
Test may only be collected and performed at testing laboratories (see below).
- If the patient’s hematocrit (HCT) is >55%, the volume of anticoagulant in the tube should be adjusted. Contact a performing location for special tube preparation to reduce the citrate in the collection tube. For those patients with HCT >65.4% see LAB3632 Stago Sodium Citrate Adjustment Chart for formula to determine amount of citrate removal required.
- When possible, use fresh venipuncture to collect blood to avoid activation of coagulation by tissue thromboplastin. Blood may be drawn from an indwelling catheter or vascular access device. Draw sample from a lumen that has not been heparinized or flush the line with 5 mL of saline, and discard or use for other purposes the first 5-10 mL of blood.
- Butterfly device collections, draw a discard tube first to remove air from collection tubing.
- Tubes should be >90% filled.
- Immediately mix gently after collection by inverting the tube end over end 5 to 6 times. Avoid vigorous mixing or additional inversion. Observe for the presence of clots. Recollect if fibrin clots observed.
- Hand-deliver to the laboratory immediately after collecting at ambient temperature.
|
SPECIMEN PROCESSING |
Specimen processing instructions: |
Deliver to laboratory immediately after collection. Do not send sample through the pneumatic tube system. |
Transport temperature: |
Room temperature |
Specimen stability: |
2 hours |
Rejection criteria: |
Clotted specimen, arterial or capillary specimen, improperly filled tube, centrifuged specimen, stability limits exceeded (2hr). Sample sent through pneumatic tube system. HCT > 55% without citrate adjusted tube. |
TEST DETAILS |
Reference interval: |
Reference ranges established using venous blood collected in sodium citrate tubes from normal adult (>18 years old) healthy donors not on anticoagulation. They do not reflect patients with different baseline coagulation values (examples: liver disease, pregnant women, children, hemophiliacs) or patient samples from a different source or collected in different specimen container.
| R |
2.5-8.3 min |
| K |
0.5-3.7 min |
| Angle |
46.8-78.4 degrees |
| MA |
50.6-72.5 mm |
| LY 30 |
0–7.5% |
| CI |
-3 to 3 |
|
Interpretation guide: |
Figure 1. Typical TEG Tracing:
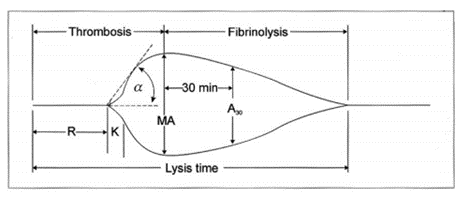
| Table 1: TEG Measured Parameters and Meanings |
| Measurement |
Definition |
R-Time
(Reaction time) |
Start of reaction to initial clot formation. Prolonged by anticoagulants (heparin, low molecular weight heparin, direct thrombin inhibitors, warfarin, etc.) and factor deficiencies. Shortened by hypercoagulable conditions. Most similar tests are PT and APTT. |
K-Time
(Kinetic time) |
Period at the end of R-time to the 20mm. Represents when fibrin strands crosslinked into fibrin meshwork. |
| Angle (alpha) |
The rate of clot formation and growth. Angle reflects fibrinogen activity. |
MA
(Maximum Amplitude) |
Maximum clot strength, which is the result of two components: fibrin (minor component) and platelets (major component). Most similar test is platelet count. |
| Coagulation Index (CI) |
Instrument calculated parameter. Represents global clotting ability. Parameter not validated and clinical usefulness unclear. |
Ly30,
(Lysis at 30 min)* |
Represents clot breakdown. Similar tests include fibrin degradation products, D-dimer, or soluble fibrin monomer. It is increased in DIC or other hyperfibrinolytic states. Values >3% may be predictive of massive transfusion in trauma patients. |
| *An abnormal TEG in a patient without clinically relevant bleeding does not require transfusion of blood components |
|
Additional information: |
TEG tracing available for real-time review using TEG Manager (requires GOAL course completion for website access). |
CPT code(s): |
85347 x2, 85384 x2, 85576 x2
|
|
Note: The billing party has sole responsibility
for CPT coding. Any questions regarding coding should be directed
to the payer being billed. The CPT codes provided by GML are
based on AMA guidelines and are for informational purposes only. |
Test includes: |
Kaolin, Kaolin + Heparinase. |
Methodology: |
Viscoelastic measurement of clot formation |
Synonyms: |
Viscoelastography, Thromboelastogram
|
Clinical significance: |
Global assessment of coagulation using whole blood that is typically utilized to guide transfusion in patients with trauma, recent surgery, or bleeding patients. There is no single ideal test to assess blood coagulation, and this should be used in conjunction with other coagulation studies and clinical findings. Limitations of the assay include:
- Tissue factor, the initiator of the coagulation cascade, cannot be quantified by this test and abnormal levels cannot be detected.
- This test does not assess the size of the injured vessel, blood flow characteristics, and local vessel wall biology, all of which affect blood coagulation.
- Reference ranges do not reflect patients with atypical baseline coagulation values (examples: liver disease, pregnant women, children, hemophiliacs) or patient samples from arterial blood or collected in different specimen container. Therefore, parameter changes and transfusion therapy recommendations in these patient populations should not solely be based on numerical results, but TEG tracings and clinical state should also be considered.
- TEG has a sensitivity and specificity that may vary significantly in different populations (pediatrics, pregnant women, liver disease, hemophiliacs) and results in these populations should be interpreted with caution.
- Anticoagulants and Antiplatelet agents may not be detected with TEG.
- Warfarin is a commonly prescribed medication. In about half of patients on warfarin therapy, R-time may be normal in TEG. Poor correlation between TEG and INR is known. TEG should NOT be used to monitor warfarin therapy, instead an INR is still the gold standard for warfarin therapy monitoring.
- To assess platelet function for patients on a P2Y12 inhibitor (clopidogrel) or aspirin a platelet function assay (VerifyNow) is recommended.
- To exclude venous thrombosis or pulmonary embolus in low-risk outpatients, a D-Dimer should be used.
- Previous studies demonstrate a significantly elevated CI in the postoperative period after general surgery and in cancer patients, suggesting a prothrombotic state. However, this index is not widely used, and its clinical usefulness is not yet validated. Patients being assessed for long-term anticoagulation should undergo appropriate advanced thrombophilia diagnostic tests as the TEG is not validated for these conditions.
- Lysis 30 reference range adopted based on literature, which showed previously healthy individuals can have lysis 30 values up to 3%. This is not a sensitive marker for fibrinolysis and values <3% do not rule out hyperfibrinolysis.
|
Doctoral Director(s:) |
Michelle Grant DO |
| Review Date: |
10/31/2023 |

